FDA’s Zero-Tolerance Standard for Pathogens
Monday, 06 April 2026
by Dr Faour Klingbeil
You are a processor exporting to the U.S. or planning to. If the Preventive Controls rule (PC rule) applies to you, then you need a compliant Food Safety Plan hashtag#fsp. And, If you’re a PCQI, you should be familiar with the 21 CFR §117.136 under the PC rule. The provision that allows a manufacturer to rely on a downstream customer to control an identified hazard, instead of implementing a preventive control at their own facility. So, does that mean you can legally ship a product with a known pathogen hazard… without controlling it? Be cautious with the interpretation. This is one of the provisions that might be misread/misinterpreted. The answer may be a “yes”, but only under tightly defined conditions. I underline “tightly defined”. Because this is true only under very specific, tightly controlled conditions. It’s a conditional regulatory pathway, built on documentation, disclosure, and accountability. When does downstream reliance apply? This is especially relevant in supply chain situations where a facility receives raw materials that may carry inherent hazards, but has no lethal step to eliminate them, e.g., raw cocoa, coffee beans, flour, grains.. In such cases, control is intentionally placed downstream, not ignored. All these conditions should me met: Does this relax zero tolerance policy for pathogens such as Salmonella? No. § 117.136 addresses a specific gap: your facility has no lethal step to eliminate hazards inherent in raw ingredients. It does not mean a facility can allow Salmonella to persist and expect the customer to clean it up. FDA expectations for Listeria monocytogenes and Salmonella do not disappear at handoff. And under the FD&C Act, food must not be adulterated, regardless of where in the chain control is applied. What this provision does NOT mean It is not a relaxation of pathogen standards.Zero-tolerance expectations for Salmonella and Listeria monocytogenes remain in full effect at every step in the chain. It is not a default option.This is a conditional, documented pathway; it applies only where your process genuinely cannot control the hazard. It is not an exemption from GMP or sanitation.You must still prevent contamination and ensure your process does not introduce or amplify hazards. It is not a transfer of legal accountability.Under the FD&C Act, food must not be adulterated; this applies regardless of where in the chain control is applied. Therefore, even when control is deferred, you must still: Control may be transferred under certain conditions, but your responsibility cannot be, and that does not mean the zero-tolerance policy is relaxed. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, Food safety and trade, FSMA
Which Facility Must Register With The FDA?
Friday, 27 March 2026
by Dr Faour Klingbeil
I often get questions on FDA registration from both startups and established companies. For example, Does our third-party warehouse need to register with FDA? If it holds food for US consumption, most likely, yes.First, what does the law state? FSMA requires that domestic and foreign facilities that manufacture, process, pack, or hold food for human or animal consumption in the U.S. must register with the FDA. So, a third-party warehouse used exclusively for storing your finished products is still a food holding facility. That brings it within FDA’s registration requirement, regardless of whether it handles products from one company or many. Almost all warehouses that handle food meet at least the “hold” threshold under FSMA, which means they are: 🔸Subject to FDA registration 🔸Expected to have applicable food safety controls in place This applies to domestic and foreign facilities alike. If the facility packs or holds food after the last foreign manufacturer or processor, the foreign packer or holder must also register. In multi-facility chains, more than one site may need registration. Exemptions apply to retail food establishments (restaurants/groceries) and farms holding their own produce are typically exempt. Pure carriers (trucks in transit) are generally not considered facilities, but the warehouses they transfer to are. ❗️NOTE: FDA registration is free See below details on which facility types must register, including scenarios involving multiple foreign facilities. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, FSMA
51% of DG SANTE Agri-Food Controls in 2026 Target Third Countries: Inside the European Commission’s Audit Programme
Saturday, 28 February 2026
by Dr Faour Klingbeil
The European Commission’s 2026 Health and Food Audits and Analysis Programme sends an important signal to global agri-food exporters. The message is clear:EU import controls are entering a new phase of intensified scrutiny. In response to rising concerns around food safety, regulatory alignment, and the integrity of third-country control systems, the EU is significantly scaling up audits of non-EU countries. The focus areas for 2026 include: 🔸 Pesticide residues🔸 Pharmacologically active substances in animal products🔸 Contaminants and mycotoxins🔸 Food contact materials (including recycled plastics)🔸 Microbiological risks🔸 Animal health and welfare standards The Commission has committed to a 50% increase in audits of non-EU countries between 2025–2027. In 2026 alone, 51% of agri-food controls will target non-EU countries, compared to 33% in 2025. For exporters to the EU market, this marks a structural recalibration of import scrutiny. The emphasis is no longer limited to product-level compliance, it extends to the robustness of official control systems in exporting countries. In times of geopolitical instability and economic uncertainty, regulatory certainty becomes even more critical. The EU is reinforcing its food safety perimeter. If your business exports to the European Union, 2026 will require: 🔸 Robust residue monitoring aligned with EU MRLs🔸 Strong and defensible official certification systems🔸 Full traceability across the supply chain🔸 A documented and demonstrable compliance culture🔸 Preparedness for on-site Commission audits Preparedness is not optional. It is strategic for market growth and business expansion. Below is the list of sectors falling under heightened European Commission scrutiny in 2026, together with the planned audits for non-EU countries. dfk DFK Safe Food Environment · Regulatory Services EU Food SafetyIncreased Audits 2026 Food categories under highest scrutiny in third-country controls, verified from the European Commission DG SANTE Health and Food Audits and Analysis Programme 2026 159 Total Controls 51% Non-EU Focus, up from 33% 150 Audits Planned ~50% Increase in non-EU audit share 2025-27 Food Categories Ranked by Planned Audit Intensity, Third Countries # Food Category and Scope Audits Relative Intensity Priority 1 Fish, Fishery Products and Fish Oil China, Chile, Oman, Panama, Türkiye, Uruguay; plus 7 desk-based audits (Papua New Guinea, Senegal, Cabo Verde, Solomon Islands, Taiwan, Nigeria, Malaysia) 13audits High 2 Residues of Pharmacologically Active Substances, Food of Animal Origin Tunisia, Panama, Bosnia and Herzegovina, Cambodia, Republic of Korea, Bangladesh, Uruguay, Türkiye, Moldova, plus 1 TBD; desk-based assessment of 30 non-EU countries 10audits High 3 Plants and Plant Products, Phytosanitary Controls Uganda, Costa Rica, China, Japan (Bonsai), Peru, Thailand, plus 1 TBD 7audits High 4 Pesticide Residues, Food of Plant Origin Kenya, Thailand, Sri Lanka, Bangladesh, Madagascar 5audits High 5 Food Contact Materials, Recycled Plastics United Kingdom, India, Türkiye, China; first coordinated audit programme on this topic launched in 2026 4audits New Topic 6 Meat and Meat Products, Beef and Poultry Brazil (beef), Argentina (poultry and wild hare), United Kingdom (beef), Uruguay (beef) 4audits Medium 7 Animal Health, Poultry Meat and Eggs China, Thailand, United States 3audits Medium 8 Live Bivalve Molluscs Japan, Vietnam 2audits Medium 9 Microbiological Safety, Food of Non-Animal Origin Serbia, Türkiye 2audits Medium 10 Contaminants, Food of Non-Animal Origin United States; Türkiye (product-specific details per Annex 4 country audit descriptions: aflatoxin in peanuts; mycotoxins in pistachios and dried figs) 2audits Medium Primary Risk Concerns Driving the 2026 Audit Programme Pesticide Residues (MRL Compliance) Veterinary Drug Residues Mycotoxins and Aflatoxins Microbiological Safety Food Contact Material Migration Animal Disease Certification Chemical Contaminants (incl. heavy metals, where relevant) Antimicrobial Resistance (AMR) Countries Facing Multiple Audit Topics in 2026 Türkiye Vet. drug residues, pesticides and contaminants Fishery product production controls Food contact materials (recycled plastic) Mycotoxins in pistachios and dried figs Microbiological safety (FNAO) Food irradiation facilities China Poultry meat and eggs, animal health Fish oil for human consumption Food contact materials (recycled plastic) Plant and plant products export controls Uruguay Fishery products Beef production controls Vet. drug residues and contaminants Bangladesh Pesticide residues, food of plant origin Vet. drug residues and contaminants Panama Fish oil for human consumption Vet. drug residues and contaminants Thailand Poultry meat and eggs, animal health Pesticide residues, food of plant origin Plant and plant products export controls New Audit Topics Launched in 2026 Fish Oil: first coordinated audit programme on this topic, covering China, Chile, Oman, Panama; EU Member States France and Germany, targeting products for human consumption. Food Contact Materials with Recycled Plastics: first coordinated audit programme on this topic in EU (Netherlands, Poland) and third countries (UK, India, Türkiye, China), addressing chemical migration risks from recycled packaging. How Audit Outcomes Affect Market Access A negative Commission audit outcome in a third country can trigger trade-restrictive measures including enhanced border checks or import suspension. Audits verify the competent authority’s official control system, not just individual product lots. Results are published on the DG SANTE audit portal and inform EU import policy decisions. Source: EC DG SANTE · Health and Food Audits and Analysis Programme 2026 · EW-01-25-098-EN-N dfk Safe Food Environment
- Published in EU regulatory compliance, Food safety and trade
When Supplier Controls Matter More Than Your Own CCPs
Monday, 16 February 2026
by Dr Faour Klingbeil
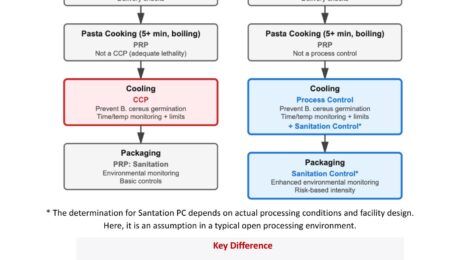
A single ingredient , pre-cooked pasta, caused 7 deaths, 28 illnesses, 27 hospitalizations and brought 6+ brands into recall. This multistate Listeria monocytogenes outbreak was linked to prepared, ready-to-eat pasta meals. We are not here to speculate about failure or assign blame. But we should reflect. Irrespective of this specific case, where the exact root cause and system robustness are not fully clear, such incidents remind us of something critical: It is time for food safety to move beyond a purely HACCP-centered mindset. HACCP asks: That logic is vital, indispensable. But it is not sufficient. When conducting risk assessments and determining management strategies, food businesses must go beyond labeling controls as CCPs or PRPs. We need a broader, risk-based Food Safety Plan mindset. In cases like this, pre-cooked, hydrated components used in RTE foods inherently require elevated scrutiny not because something necessarily failed, but because the risk profile demands it. A broader system means: • Strong supply-chain preventive controls for high-risk ingredients.HACCP often treats supplier controls as PRPs. A Food Safety Plan treats high-risk incoming ingredients as Supply Chain Preventive Controls, applying CCP-equivalent rigor. • Sanitation Preventive Controls when the hazard is environmentalWhen environmental contamination is a reasonably foreseeable hazard, sanitation is no longer “just a PRP”, it becomes a managed preventive control. • Allergen Preventive Controls where cross-contact risk is significant.When severity is high, allergen management requires structured and robust preventive controls. • Verification beyond documentation.Does your supplier have validated preventive controls?Are you reviewing real performance data and environmental monitoring trends — not just Certificates of Analysis? • Environmental monitoring expectations at supplier level, where relevant. • Risk-based intensity of oversight proportional to severity. • and more…. Food Safety Plans change how we think. They shift us from diagram compliance to hazard-control strategy design. And that shift is not theoretical. It is necessary. If you manufacture or brand RTE foods, your strongest critical control may ALSO sit at your supplier’s site. I have prepared a very simplified illustration showing how HACCP and a Food Safety Plan approach may differ, both at the level of the pre-cooked pasta manufacturer and at the level of the brand owner using it in finished RTE products. Have a look at it here: If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, Food safety and trade, FSMA
Tagged under:
#foodsafetyplan, #fsp, #HACCP, #riskmanagement, Food Safety, Regulatory compliance, Risk assessment
EU Compliance Is Not FDA Compliance: What Ingredient Status Means for US Market Access
Thursday, 29 January 2026
by Dr Faour Klingbeil
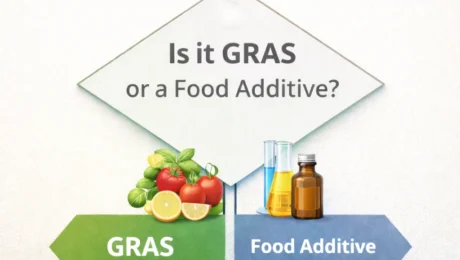
If raw materials aren’t fully FDA-compliant but EU compliant, can we still sell the product in the US ? This question comes up often with complex formulations. From an FDA perspective, the logic is simple. Pointing to approval in other jurisdictions, relying on a long history of use elsewhere, or assuming that a low use level or a so-called “minor” ingredient will be overlooked is not a good argument or approach to access the US market. These factors may feel reassuring, but they do not establish legal status under FDA rules. What does work is a disciplined regulatory approach by mapping each ingredient and sub-ingredient to its US regulatory status, verifying that the intended use levels and conditions of use are covered, and reformulating where no valid approval or GRAS pathway exists. I have prepared this illustration that summarizes the decision logic used to determine a substance’s regulatory and legal status, and shows where compliance ultimately leads to the need for food additive authorization. If you find it useful and choose to share it, attribution would be appreciated. If you have questions, feel free to reach out anytime. We support food businesses with food labeling compliance, risk assessment and management, and food system compliance with the FDA. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, FSMA
COVID-19 Preparedness in the Food Industry
Monday, 14 October 2024
by Dr Faour Klingbeil
On 11 March 2020, the outbreak of coronavirus disease (COVID-19) has been declared a global pandemic by the World Health Organization as the virus has spread to many countries. It’s the first time the WHO has called an outbreak a pandemic since the H1N1 “swine flu” in 2009. What is confirmed is that the virus is transmitted through direct contact with respiratory droplets of an infected person (generated through coughing and sneezing). Individuals can also be infected from and touching surfaces contaminated with the virus and touching their face (e.g., eyes, nose, mouth). Besides, the COVID-19 virus may survive on surfaces for several hours, yet disinfectants can kill it. On 9 March, the European Food Safety Agency (EFSA) stated on their website that there is currently no evidence that food is a likely source or route of transmission of the novel coronavirus, and that they are closely monitoring the situation as any new information about the outbreak comes to light. EFSA’s opinion is based on the fact that previous outbreaks of related coronaviruses, such as severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV), show that transmission through food consumption did not occur. BfR, the federal institute of risk assessment in Germany concurred with the findings, stating that there are currently no cases that have shown any evidence of humans being infected with the new type of coronavirus by another method, such as via the consumption of contaminated food or via imported toys. Transmission via surfaces which have recently been contaminated with viruses is, nonetheless, possible through smear infections. This is only likely to occur during a short period after contamination, due to the relatively low stability of coronaviruses in the environment. This virus is not SARS, it’s not MERS, and it’s not influenza. It is a unique virus with unique characteristics WHO Director General A recent review analyzed 22 studies and revealed that human coronaviruses such as Severe Acute Respiratory Syndrome (SARS) coronavirus, Middle East Respiratory Syndrome (MERS) coronavirus or endemic human coronaviruses (HCoV) can persist on inanimate surfaces like metal, glass or plastic for up to 9 days, but can be efficiently inactivated by surface disinfection procedures with 62–71% ethanol, 0.5% hydrogen peroxide or 0.1% sodium hypochlorite within 1 minute. Other biocidal agents such as 0.05–0.2% benzalkonium chloride or 0.02% chlorhexidine digluconate are less effective This is what we know “so far”. As the WHO Director-General stated “This virus is not SARS, it’s not MERS, and it’s not influenza. It is a unique virus with unique characteristics”, and scientists are working around the clock to address critical gaps in knowledge. In a recent study (US government work) conducted by the National Institutes of Health, Princeton University and the University of California, Los Angeles, with funding from the U.S. government and the National Science Foundation, Covid-19 was detected up to three hours later in the air, up to four hours on copper, up to 24 hours on cardboard and up to two to three days on plastic and stainless steel. Revise and update the health policy Having said that, while there is a lot of uncertainty in the situation, it is advisable that food processors evaluate the current practices inside their organization and manage staff who may be carriers or infected with the Coronavirus. Reviewing and updating the existing disease control/health policy while considering recent recommendations and requirements of the local authorities is paramount. Did you address in your your policies address any food to which an ill employee may have had exposure, including whether the policies address whether there are conditions present that would support a company decision to place food on hold pending advice from the public health authorities. Here are the CDC recommended strategies for employers to use which may help may help prevent workplace exposures to acute respiratory illnesses, including COVID-19. Many of the steps described in the CDC link are useful and apply to every food facility, yet as a food facility outside the US, you will refer to the guidance, updates and travelers’ health notices of local authorities to determine whether there are any local requirements to contact public health authorities in the event an employee in the workplace has been diagnosed with COVID-19 and to establish procedures for handling other employees who may have come in contact with the diagnosed employee. Top management should encourage employees with symptoms to stay home and get prepared for the sudden absenteeism and shortage of staff. Besides the disease control plan, precautionary hygiene measures are of significant importance. The World Health Organisation (WHO) has issued recommendations including advice on following good hygiene practices during food handling and preparation, such as washing hands, cooking meat thoroughly and avoiding potential cross-contamination between cooked and uncooked foods. Similarly, many local authorities such as in Belgium also emphasized those practices. As simple as this seems to be, translating messages into practices is not easy and often undermined with barriers that need to be understood. In practice, it requires instilling a hygiene culture to ensure the highest levels of hygiene measures, particularly when food processors may face the situation of operating with staff shortages and absenteeism. A hygiene culture in a way it does not require the staff to think much about it! it is a culture shared at all levels of the organization from top management to production staff. Have you included in your contingency plan re-evaluating your hygiene standards? The topic of today is focused on hand hygiene, which must not be overlooked in your Covid-19 training tool box. What have you done to highlight this issue in your organization? It would be great if you share your experience with others in the comment box. I am sharing some of the key messages you may like to consider to reinforce the hand washing practices: Hygiene Culture 1- Assign competent staff to stay aware of recent news and updates on the COVID-19 outbreak, how it is transmitted and how to prevent transmission. Information are available on the CDC, ECDC, WHO websites, and local health authorities.
- Published in Covid-19
The decision of when and which validation study approach to adopt
Friday, 24 March 2023
by Dr Faour Klingbeil
The PCQI course participants often showed a particular interest in the topic of validation, when and how to validate, what is acceptable and what is not, with regards to compliance with the FDA requirements. The Preventive Controls for Human Food requirements stipulate in 21 CFR 117.160: (a) You must validate that the preventive controls identified and implemented in accordance with § 117.135 are adequate to control the hazard as appropriate to the nature of the preventive control and its role in the facility’s food safety system. (b) The validation of the preventive controls must be performed (or overseen) by a preventive controls qualified individual. According to § 117.3 Definitions, validation means obtaining and evaluating scientific and technical evidence that a control measure, combination of control measures, or the food safety plan as a whole, when properly implemented, is capable of effectively controlling the identified hazards. While the Preventive Controls for Human rule provided flexibility in justifying the reasons not to validate sanitation preventive controls, allergen preventive controls, and others – except in certain conditions (to be discussed in the next post), validation of process controls is a must-to-do. This decision tree , published by Ceylan et al. 2021 in Comprehensive Reviews in Food Science and Food Safety, is a practical and simple tool to use in supporting the decision of when and which validation study approach is most applicable. You find the decision tree in the link provided above to access the full article. If you find it useful, why not sharing it with you team members to enhance their knowledge on the importance of science-based approach in food safety. Got questions on your FSP and risk assessment, or on regulatory matters? I will be happy to hear from you.
- Published in FDA Compliance, Food safety and trade, FSMA
The All-Too-Human Causes of Food Safety System Shortfalls
Sunday, 29 August 2021
by Dr Faour Klingbeil
Moving from knowledge-based to behavior-based training might be key to culture change An article published in the Food Safety Magazine, August 12, 2021 Given the continued growth of trade agreements and exchanges between countries and the evolution of production methods to meet international market needs, our food supply has developed significantly over the last several decades. A wide array of our food products are made from ingredients and packaging sourced from different suppliers worldwide, resulting in rapid movements of food products and globalized food transport. The “international agro-food trade network,” constructed using the United Nations (UN)’s food-trade data, shows the dense web of food trade connections among seven central countries that trade with more than 77 percent of the 207 countries from which the UN gathers information.1 While this vast trade network enhances accessibility to food, considerable risks emerge with the amplified production and intensive handling of raw materials across the supply chain, further complicating the tracing of food sources or foodborne hazards in multiple actors’ global supply chain. Given the continued growth of trade agreements and exchanges between countries and the evolution of production methods to meet international market needs, our food supply has developed significantly over the last several decades. A wide array of our food products are made from ingredients and packaging sourced from different suppliers worldwide, resulting in rapid movements of food products and globalized food transport. The “international agro-food trade network,” constructed using the United Nations (UN)’s food-trade data, shows the dense web of food trade connections among seven central countries that trade with more than 77 percent of the 207 countries from which the UN gathers information.1 While this vast trade network enhances accessibility to food, considerable risks emerge with the amplified production and intensive handling of raw materials across the supply chain, further complicating the tracing of food sources or foodborne hazards in multiple actors’ global supply chain. Indeed, a loss of control or oversight at any step of the supply chain could lead to detrimental economic and public health consequences. In 2011, one of the largest outbreaks of a foodborne illness was caused by enterohemorrhagic Escherichia coli O104:H4. The deadly strain caused approximately 3,000 hospitalized cases, 855 of them due to hemolytic uremic syndrome. It also led to 55 deaths, primarily in Germany, with scattered cases in 15 other countries in Europe and North America. As the strain source was still unknown, the blame was falsely directed at Spanish cucumbers and tomatoes. Consequently, a Russian ban on imports of all European Union fresh produce, followed by the EU’s ban on the import and sale of fenugreek seeds, which was eventually shown to be the culprit, caused substantial economic losses to farmers and industries.2 Such an outbreak demonstrates how local infection agents can bring about widespread economic and health threat. “Food safety systems are vital to control food safety risks, but they are not a silver bullet.” Dima Faour-Klingbeil Understanding Global Challenges Inadequate Food Safety Management Systems The global food market’s rising challenges rationalize the strict measures the food industry should take and the urgency to adopt stringent risk-based preventive food safety standards to minimize the health risks associated with consuming unsafe food products. Introduced in the 1960s by the U.S. National Aeronautics and Space Administration, the Hazard Analysis and Critical Control Points (HACCP) preventive approach was further developed by the food industry (i.e., Pillsbury). Later on, HACCP was advocated and promulgated by international organizations and mandated by regulatory agencies such as the U.S. Food and Drug Administration as an effective preventive tool to manage the hazards throughout the farm-to-fork continuum and reduce the risks associated with foodborne diseases. At the same time, driven by legal obligations to exercise due diligence and safe food production, industry stakeholders, such as retailers and nonprofit organizations, developed voluntary private food safety standards that integrate the HACCP system advocated by Codex Alimentarius to protect the reputations of businesses. Although voluntary, they generally became de facto mandatory standards that set out requirements for a risk-based food safety management system that were stricter than regulatory standards. Progressively, they were established to ensure compliance with customers’ demands and regulatory requirements while addressing fraud and intentional adulteration in a global market. Food safety systems are vital to control food safety risks, but they are not a silver bullet. Despite improvements in prevention systems, food recalls and illness outbreaks continue to hit the headlines, sometimes caused by food companies that passed certification audits of their food safety systems. A case in point is the massive multistate Salmonella outbreak caused by Peanut Corporation of America, which reportedly scored high on a third-party certification audit report.3 Read all the article in the Food Safety Magazine
- Published in Food safety and trade
Temporary COVID-19 Policy for Receiving Facilities in Meeting FSMA Supplier Verification Onsite Audit Requirements
Wednesday, 14 October 2020
by Dr Faour Klingbeil
As the global pandemic of COVID-19 continues to bring about turmoil and health threats, the U.S. Food and Drug Administration issued guidance to communicate the temporarily drop of the supplier verification onsite audit requirements for receiving facilities and importers under the FDA Food Safety Modernization Act (FSMA) provided other supplier verification methods are used instead. The Preventive Controls for Human Food (PC Human Food) rule requires receiving facilities to conduct supplier verification activities based on the hazard analysis conducted as part of their written Food Safety Plan. These verification activities generally include onsite audits, sampling and testing, or a review of food safety records. Receiving facilities may determine onsite audits to be the most appropriate supplier verification activity. However, many governments have taken strict measures to limit unnecessary inland and out-land travels in an effort to curb the spread of the COVID-19 coronavirus. Such much needed restrictions may impact the ability of receiving facilities to conduct or obtain onsite audits of their suppliers. Therefore, the guidance outlines the circumstances under which FDA does not intend to enforce the requirement to conduct or obtain an onsite audit of a food supplier when the food supplier is in a country or region covered by a government travel restriction or advisory related to COVID-19. The FDA anticipates that receiving facilities will resume onsite audits within a reasonable period of time after it becomes practicable to do so, and update their food safety plans accordingly. FDA intends to provide timely notice before withdrawing this policy. Specifically, FDA does not intend to enforce the requirement foran onsite audit in the following circumstances: A receiving facility has determined that an onsite audit is the appropriate verification activity for an approved supplier, as reflected by its written food safety plan or FSVP; The supplier that is due for an onsite audit is in a region or country covered by a government travel restriction or travel advisory related to COVID-19; Because of the travel restriction or travel advisory, it is temporarily impracticable for the receiving facility to conduct or obtain the onsite audit of the supplier; and The receiving facility temporarily selects an alternative verification activity or activities, such as sampling and testing food or reviewing relevant food safety records, and modifies its food safety plan to incorporate the alternative activity or activities. The alternative verification activity or activities are designed to provide temporary assurance that the hazard requiring a supply-chain-applied control has been significantly minimized or prevented during the period of onsite audit delay. FDA anticipates that receiving facilities will resume onsite audits within a reasonable period of time after it becomes practicable to do so, and update their food safety plans accordingly. FDA intends to provide timely notice before withdrawing this policy. Sources: The Center for Food Safety and Applied Nutrition Constituent Update
- Published in Covid-19
Microbiological quality of ready-to-eat fresh vegetables and their link to food safety environment and handling practices in restaurants in Lebanon
Thursday, 08 October 2020
by Dr Faour Klingbeil
The increased consumption of ready-to-eat (RTE) salads outside homes as a result of a fast paced lifestyle, awareness on their nutritional attributes and enhanced processing technology is well documented. Outbreak investigations often indicate that food service establishments greatly contribute to food-borne illnesses involving fresh produce. Fifty small and medium sized (SME) restaurants in Beirut were surveyed for their food safety climates. A total of 118 samples fresh-cut RTE salads vegetables and 49 swabs of knives and cutting boards were collected for microbiological analysis. A number of food safety practices concerns were identified in this study. The general lack of cleaning and sanitization procedures combined with a clear evidence of cross-contamination opportunities were generally reflected in the overall unsatisfactory quality of RTE vegetables. The majority of SMEs were unaware of the significance of applying control measures when handling vegetables and of the fundamental requirements for separate sinks used for hand-washing and washing vegetables. The inappropriate sanitation measures were not applied in 60% of the premises and a large percentage of food businesses (64%) lacked hand-washing sinks. A large proportion (84%) reported that the wash water was neither treated nor filtered and did not use sanitizers. More than half of the RTE salad vegetables were unsatisfactory due to E. coli and Listeria spp. counts that exceeded the criteria limits >100 CFU/g indicating poor hygienic practices and sanitary conditions. High frequency of S. aureus was also observed indicating poor hygiene practices of food handlers. Presence of Listeria monocytogenes and Salmonella spp. were traced back to samples obtained from restaurant that had no hand-washing sinks, fresh vegetables washing sinks, and adequate preparation and storage areas; the corresponding inspection rating recorded 32 over 100 possible points. The high microbial population size on chopping board surfaces offered an additional assumption for the actual contamination levels observed on RTE vegetables. As the correlation between the total inspection scores and the microbiological indicators were found not significant, our study confirmed that the total inspection scores per se would not directly indicate the microbiological safety of RTE vegetables in the restaurants and that the strategy of end-product testings do not and will not provide safe vegetables to consumers. Interestingly, there were variations in microbial counts and a significant correlation of high Listeria levels with individual inspection components, i.e., the inadequate cleaning and poor cross-contamination preventive measures, which emphasized that shortfalls in those particular points in the processing environment possibly indicate the presence of pathogens, e.g., L. monocytogenes on fresh vegetables. Therefore, the applications of critical control points for the preparation of fresh salad vegetables and personnel training on the associated bacterial hazards are fundamentals, particularly when salads are prepared in small working facilities in SMEs. The high microbial loads in RTE vegetables found in this work serve as an indicator for the need to promote awareness and a guidance for local authorities on the critical areas commonly identified in the SMEs that most likely affect the safety of fresh vegetables. It underscored the vigilant cleaning and sanitation procedures to reduce or eliminate contamination and cross-contamination risks that may occur at the pre-farm gate and throughout the supply chain stages.
- Published in Ready to eat vegetables
- 1
- 2