Which Facility Must Register With The FDA?
Friday, 27 March 2026
by Dr Faour Klingbeil
I often get questions on FDA registration from both startups and established companies. For example, Does our third-party warehouse need to register with FDA? If it holds food for US consumption, most likely, yes.First, what does the law state? FSMA requires that domestic and foreign facilities that manufacture, process, pack, or hold food for human or animal consumption in the U.S. must register with the FDA. So, a third-party warehouse used exclusively for storing your finished products is still a food holding facility. That brings it within FDA’s registration requirement, regardless of whether it handles products from one company or many. Almost all warehouses that handle food meet at least the “hold” threshold under FSMA, which means they are: 🔸Subject to FDA registration 🔸Expected to have applicable food safety controls in place This applies to domestic and foreign facilities alike. If the facility packs or holds food after the last foreign manufacturer or processor, the foreign packer or holder must also register. In multi-facility chains, more than one site may need registration. Exemptions apply to retail food establishments (restaurants/groceries) and farms holding their own produce are typically exempt. Pure carriers (trucks in transit) are generally not considered facilities, but the warehouses they transfer to are. ❗️NOTE: FDA registration is free See below details on which facility types must register, including scenarios involving multiple foreign facilities. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, FSMA
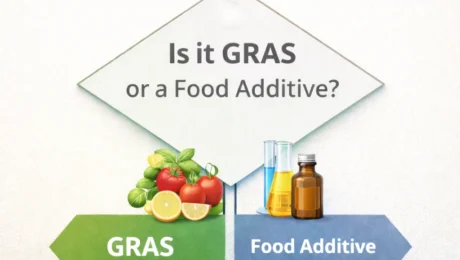
EU Compliance Is Not FDA Compliance: What Ingredient Status Means for US Market Access
Thursday, 29 January 2026
by Dr Faour Klingbeil
If raw materials aren’t fully FDA-compliant but EU compliant, can we still sell the product in the US ? This question comes up often with complex formulations. From an FDA perspective, the logic is simple. Pointing to approval in other jurisdictions, relying on a long history of use elsewhere, or assuming that a low use level or a so-called “minor” ingredient will be overlooked is not a good argument or approach to access the US market. These factors may feel reassuring, but they do not establish legal status under FDA rules. What does work is a disciplined regulatory approach by mapping each ingredient and sub-ingredient to its US regulatory status, verifying that the intended use levels and conditions of use are covered, and reformulating where no valid approval or GRAS pathway exists. I have prepared this illustration that summarizes the decision logic used to determine a substance’s regulatory and legal status, and shows where compliance ultimately leads to the need for food additive authorization. If you find it useful and choose to share it, attribution would be appreciated. If you have questions, feel free to reach out anytime. We support food businesses with food labeling compliance, risk assessment and management, and food system compliance with the FDA. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, FSMA
DFK Supports SMEs and Stakeholders Through the USAID TRADE Project on Export Enhancement Program
Tuesday, 26 March 2024
by Dr Faour Klingbeil
As part of the Export Enhancement Program under the USAID TRADE project, in collaboration with the Food Export Council (FEC), DFK has been dedicated to supporting SMEs and key stakeholders by enhancing their knowledge and capabilities. This initiative, which began in September 2022, continued through multiple training sessions held on: The objective of these sessions was to train and build the capacity of industry delegates and trainers on the FDA requirements under the FSMA preventive controls rule key and topics essential for international trade compliance and food safety, including: This effort continued through 2024 with the development of Trainer of Trainers (ToT) guidance documents, including: Through this project, DFK is empowering SMEs with the critical knowledge and skills needed to meet international standards, fostering safer food products and enhancing export capabilities.
DFK Continues Its Collaboration with the Food Export Council, Delivering Guidance on FDA FSMA and Traceability RequirementsJuly 2023 – Cairo, Egypt
Sunday, 23 July 2023
by Dr Faour Klingbeil
For the second consecutive year, DFK has strengthened its collaboration with the Food Export Council (FEC) to support Egyptian food manufacturers and exporters in meeting international regulatory requirements. As part of this ongoing partnership, Dr. Dima Faour-Klingbeil delivered a webinar in July focused on the U.S. Food Safety Modernization Act (FSMA) and the FDA’s Food Traceability Rule. The webinar organized and hosted by the FEC provided exporters with essential guidance for accessing and maintaining compliance in the U.S. food market. Dr. Faour-Klingbeil outlined the transformative nature of FSMA, the most comprehensive reform of U.S. food safety law in decades, which shifts the regulatory system from reacting to food safety incidents to preventing them. During the session, she explained FSMA’s core principles, including preventive controls, inspection and compliance mechanisms, import oversight, and the FDA’s authority to act swiftly on products suspected of contamination. She highlighted key rules that foreign suppliers must understand and especially the Foreign Supplier Verification Program (FSVP), which requires U.S. importers to verify that their foreign suppliers meet U.S. standards. A major focus of the webinar was the FDA’s Food Traceability Rule, which expands traceability requirements across the global food supply chain. Dr. Faour-Klingbeil detailed the obligations for documenting Key Data Elements (KDEs) and Critical Tracking Events (CTEs), underscoring the rule’s significance for high-risk foods and the need for robust record-keeping systems to enhance recall efficiency and supply-chain transparency. Dr. Faour-Klingbeil urged Egyptian exporters to proactively strengthen their compliance systems to avoid border rejections and improve their competitiveness in the U.S. FEC emphasized that these training initiatives form part of a broader strategy to expand export capabilities and support companies participating in major global trade events. For more information, Watch the webinar in this link:
FEC Webinar on the FSMA rules and the FDA requirements for food export – Egypt
Wednesday, 25 January 2023
by Dr Faour Klingbeil
Dr Faour-Klingbeil offered a presentation on the FSMA rules and the FDA Requirements for Food Export during a webinar organized by the Food Export Council in Egypt on 18.01.2023 and attended by more than 95 participants. What are the FSMA rules? how does FSMA affects foreign facilities? what are the steps to meet the FDA requirements for food export? What is the responsibility of the PCQI? Watch here:
FAFS Lecture Series | An overview of the FSMA and its implications for food exporters
Wednesday, 16 December 2020
by Dr Faour Klingbeil
As part of the American University of Beirut FAFS (The Faculty of Agricultural and Food Sciences ) Lecture Series, Dr. Dima was invited to deliver an open lecture on “An overview of the Food and Drug Administration Food Safety Modernization Act (FSMA) and its implications for food exporters” that took place on the 10th of October 2019. About the lecture: Foodborne disease is a significant burden worldwide. In the United States, it is estimated that 48 million people get sick, 128 000 are hospitalized, and 3000 die annually from contaminated food or beverages. Prompted by the increasing prevalence of imports, high-profile food outbreaks, food fraud, and emerging hazards, the U.S. Food Safety Modernization Act was signed into law in 2011, the first historical reform to the legislative landscape of the food and beverage industry in over 70 years. FSMA has made fundamental changes to U.S. food laws. It shifted the focus of food safety from reactive to preventive approach and gave the U.S. Food and Drug Administration explicit authority and broader prevention mandate to protect consumers’ health. Instead of relying only on port-of-entry inspection, FSMA emphasizes key regulations that span across the food supply chain. Among other modifications, it imposes new responsibilities on food importers to verify that their foreign suppliers of food for human and animal consumption meet applicable FDA safety standards to ensure the safety of food imported into the U.S.