I often get questions on FDA registration from both startups and established companies. For example,
Does our third-party warehouse need to register with FDA?
If it holds food for US consumption, most likely, yes.First, what does the law state?
FSMA requires that domestic and foreign facilities that manufacture, process, pack, or hold food for human or animal consumption in the U.S. must register with the FDA.
So, a third-party warehouse used exclusively for storing your finished products is still a food holding facility. That brings it within FDA’s registration requirement, regardless of whether it handles products from one company or many.
Almost all warehouses that handle food meet at least the “hold” threshold under FSMA, which means they are:
🔸Subject to FDA registration
🔸Expected to have applicable food safety controls in place
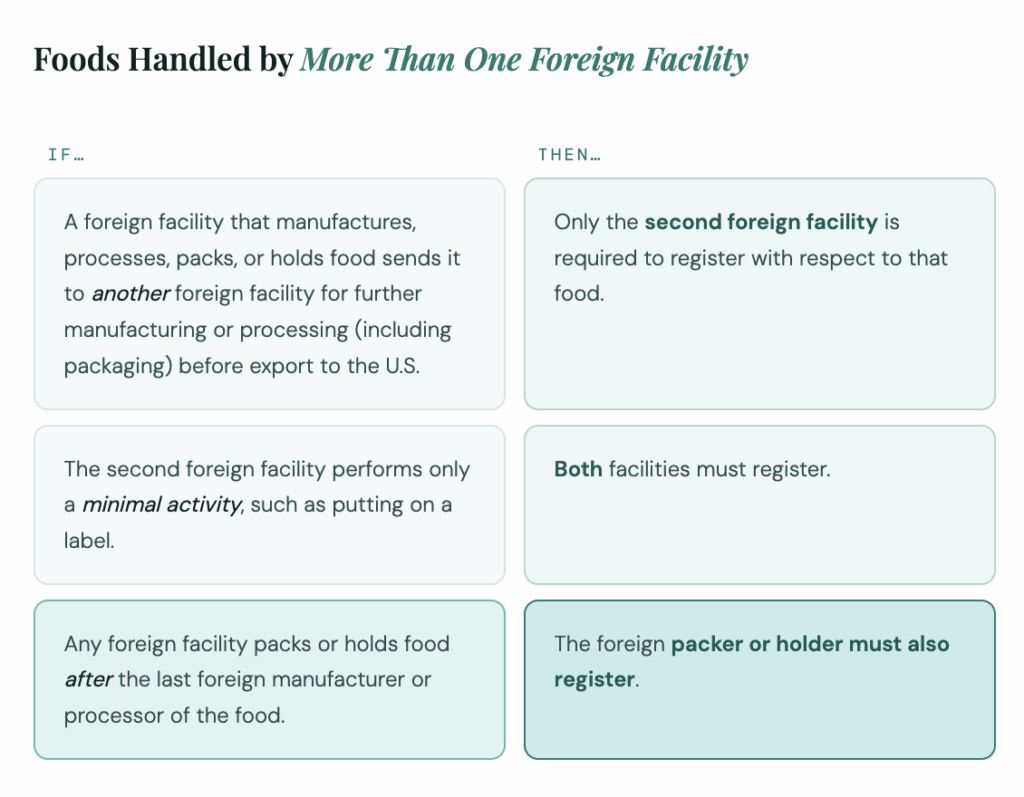
This applies to domestic and foreign facilities alike. If the facility packs or holds food after the last foreign manufacturer or processor, the foreign packer or holder must also register. In multi-facility chains, more than one site may need registration.
Exemptions apply to retail food establishments (restaurants/groceries) and farms holding their own produce are typically exempt. Pure carriers (trucks in transit) are generally not considered facilities, but the warehouses they transfer to are.
❗️NOTE: FDA registration is free
See below details on which facility types must register, including scenarios involving multiple foreign facilities.