FDA’s Zero-Tolerance Standard for Pathogens
Monday, 06 April 2026
by Dr Faour Klingbeil
You are a processor exporting to the U.S. or planning to. If the Preventive Controls rule (PC rule) applies to you, then you need a compliant Food Safety Plan hashtag#fsp. And, If you’re a PCQI, you should be familiar with the 21 CFR §117.136 under the PC rule. The provision that allows a manufacturer to rely on a downstream customer to control an identified hazard, instead of implementing a preventive control at their own facility. So, does that mean you can legally ship a product with a known pathogen hazard… without controlling it? Be cautious with the interpretation. This is one of the provisions that might be misread/misinterpreted. The answer may be a “yes”, but only under tightly defined conditions. I underline “tightly defined”. Because this is true only under very specific, tightly controlled conditions. It’s a conditional regulatory pathway, built on documentation, disclosure, and accountability. When does downstream reliance apply? This is especially relevant in supply chain situations where a facility receives raw materials that may carry inherent hazards, but has no lethal step to eliminate them, e.g., raw cocoa, coffee beans, flour, grains.. In such cases, control is intentionally placed downstream, not ignored. All these conditions should me met: Does this relax zero tolerance policy for pathogens such as Salmonella? No. § 117.136 addresses a specific gap: your facility has no lethal step to eliminate hazards inherent in raw ingredients. It does not mean a facility can allow Salmonella to persist and expect the customer to clean it up. FDA expectations for Listeria monocytogenes and Salmonella do not disappear at handoff. And under the FD&C Act, food must not be adulterated, regardless of where in the chain control is applied. What this provision does NOT mean It is not a relaxation of pathogen standards.Zero-tolerance expectations for Salmonella and Listeria monocytogenes remain in full effect at every step in the chain. It is not a default option.This is a conditional, documented pathway; it applies only where your process genuinely cannot control the hazard. It is not an exemption from GMP or sanitation.You must still prevent contamination and ensure your process does not introduce or amplify hazards. It is not a transfer of legal accountability.Under the FD&C Act, food must not be adulterated; this applies regardless of where in the chain control is applied. Therefore, even when control is deferred, you must still: Control may be transferred under certain conditions, but your responsibility cannot be, and that does not mean the zero-tolerance policy is relaxed. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, Food safety and trade, FSMA
Which Facility Must Register With The FDA?
Friday, 27 March 2026
by Dr Faour Klingbeil
I often get questions on FDA registration from both startups and established companies. For example, Does our third-party warehouse need to register with FDA? If it holds food for US consumption, most likely, yes.First, what does the law state? FSMA requires that domestic and foreign facilities that manufacture, process, pack, or hold food for human or animal consumption in the U.S. must register with the FDA. So, a third-party warehouse used exclusively for storing your finished products is still a food holding facility. That brings it within FDA’s registration requirement, regardless of whether it handles products from one company or many. Almost all warehouses that handle food meet at least the “hold” threshold under FSMA, which means they are: 🔸Subject to FDA registration 🔸Expected to have applicable food safety controls in place This applies to domestic and foreign facilities alike. If the facility packs or holds food after the last foreign manufacturer or processor, the foreign packer or holder must also register. In multi-facility chains, more than one site may need registration. Exemptions apply to retail food establishments (restaurants/groceries) and farms holding their own produce are typically exempt. Pure carriers (trucks in transit) are generally not considered facilities, but the warehouses they transfer to are. ❗️NOTE: FDA registration is free See below details on which facility types must register, including scenarios involving multiple foreign facilities. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, FSMA
51% of DG SANTE Agri-Food Controls in 2026 Target Third Countries: Inside the European Commission’s Audit Programme
Saturday, 28 February 2026
by Dr Faour Klingbeil
The European Commission’s 2026 Health and Food Audits and Analysis Programme sends an important signal to global agri-food exporters. The message is clear:EU import controls are entering a new phase of intensified scrutiny. In response to rising concerns around food safety, regulatory alignment, and the integrity of third-country control systems, the EU is significantly scaling up audits of non-EU countries. The focus areas for 2026 include: 🔸 Pesticide residues🔸 Pharmacologically active substances in animal products🔸 Contaminants and mycotoxins🔸 Food contact materials (including recycled plastics)🔸 Microbiological risks🔸 Animal health and welfare standards The Commission has committed to a 50% increase in audits of non-EU countries between 2025–2027. In 2026 alone, 51% of agri-food controls will target non-EU countries, compared to 33% in 2025. For exporters to the EU market, this marks a structural recalibration of import scrutiny. The emphasis is no longer limited to product-level compliance, it extends to the robustness of official control systems in exporting countries. In times of geopolitical instability and economic uncertainty, regulatory certainty becomes even more critical. The EU is reinforcing its food safety perimeter. If your business exports to the European Union, 2026 will require: 🔸 Robust residue monitoring aligned with EU MRLs🔸 Strong and defensible official certification systems🔸 Full traceability across the supply chain🔸 A documented and demonstrable compliance culture🔸 Preparedness for on-site Commission audits Preparedness is not optional. It is strategic for market growth and business expansion. Below is the list of sectors falling under heightened European Commission scrutiny in 2026, together with the planned audits for non-EU countries. dfk DFK Safe Food Environment · Regulatory Services EU Food SafetyIncreased Audits 2026 Food categories under highest scrutiny in third-country controls, verified from the European Commission DG SANTE Health and Food Audits and Analysis Programme 2026 159 Total Controls 51% Non-EU Focus, up from 33% 150 Audits Planned ~50% Increase in non-EU audit share 2025-27 Food Categories Ranked by Planned Audit Intensity, Third Countries # Food Category and Scope Audits Relative Intensity Priority 1 Fish, Fishery Products and Fish Oil China, Chile, Oman, Panama, Türkiye, Uruguay; plus 7 desk-based audits (Papua New Guinea, Senegal, Cabo Verde, Solomon Islands, Taiwan, Nigeria, Malaysia) 13audits High 2 Residues of Pharmacologically Active Substances, Food of Animal Origin Tunisia, Panama, Bosnia and Herzegovina, Cambodia, Republic of Korea, Bangladesh, Uruguay, Türkiye, Moldova, plus 1 TBD; desk-based assessment of 30 non-EU countries 10audits High 3 Plants and Plant Products, Phytosanitary Controls Uganda, Costa Rica, China, Japan (Bonsai), Peru, Thailand, plus 1 TBD 7audits High 4 Pesticide Residues, Food of Plant Origin Kenya, Thailand, Sri Lanka, Bangladesh, Madagascar 5audits High 5 Food Contact Materials, Recycled Plastics United Kingdom, India, Türkiye, China; first coordinated audit programme on this topic launched in 2026 4audits New Topic 6 Meat and Meat Products, Beef and Poultry Brazil (beef), Argentina (poultry and wild hare), United Kingdom (beef), Uruguay (beef) 4audits Medium 7 Animal Health, Poultry Meat and Eggs China, Thailand, United States 3audits Medium 8 Live Bivalve Molluscs Japan, Vietnam 2audits Medium 9 Microbiological Safety, Food of Non-Animal Origin Serbia, Türkiye 2audits Medium 10 Contaminants, Food of Non-Animal Origin United States; Türkiye (product-specific details per Annex 4 country audit descriptions: aflatoxin in peanuts; mycotoxins in pistachios and dried figs) 2audits Medium Primary Risk Concerns Driving the 2026 Audit Programme Pesticide Residues (MRL Compliance) Veterinary Drug Residues Mycotoxins and Aflatoxins Microbiological Safety Food Contact Material Migration Animal Disease Certification Chemical Contaminants (incl. heavy metals, where relevant) Antimicrobial Resistance (AMR) Countries Facing Multiple Audit Topics in 2026 Türkiye Vet. drug residues, pesticides and contaminants Fishery product production controls Food contact materials (recycled plastic) Mycotoxins in pistachios and dried figs Microbiological safety (FNAO) Food irradiation facilities China Poultry meat and eggs, animal health Fish oil for human consumption Food contact materials (recycled plastic) Plant and plant products export controls Uruguay Fishery products Beef production controls Vet. drug residues and contaminants Bangladesh Pesticide residues, food of plant origin Vet. drug residues and contaminants Panama Fish oil for human consumption Vet. drug residues and contaminants Thailand Poultry meat and eggs, animal health Pesticide residues, food of plant origin Plant and plant products export controls New Audit Topics Launched in 2026 Fish Oil: first coordinated audit programme on this topic, covering China, Chile, Oman, Panama; EU Member States France and Germany, targeting products for human consumption. Food Contact Materials with Recycled Plastics: first coordinated audit programme on this topic in EU (Netherlands, Poland) and third countries (UK, India, Türkiye, China), addressing chemical migration risks from recycled packaging. How Audit Outcomes Affect Market Access A negative Commission audit outcome in a third country can trigger trade-restrictive measures including enhanced border checks or import suspension. Audits verify the competent authority’s official control system, not just individual product lots. Results are published on the DG SANTE audit portal and inform EU import policy decisions. Source: EC DG SANTE · Health and Food Audits and Analysis Programme 2026 · EW-01-25-098-EN-N dfk Safe Food Environment
- Published in EU regulatory compliance, Food safety and trade
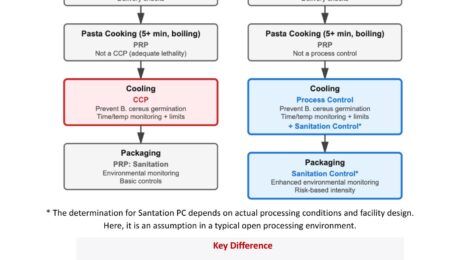
When Supplier Controls Matter More Than Your Own CCPs
Monday, 16 February 2026
by Dr Faour Klingbeil
A single ingredient , pre-cooked pasta, caused 7 deaths, 28 illnesses, 27 hospitalizations and brought 6+ brands into recall. This multistate Listeria monocytogenes outbreak was linked to prepared, ready-to-eat pasta meals. We are not here to speculate about failure or assign blame. But we should reflect. Irrespective of this specific case, where the exact root cause and system robustness are not fully clear, such incidents remind us of something critical: It is time for food safety to move beyond a purely HACCP-centered mindset. HACCP asks: That logic is vital, indispensable. But it is not sufficient. When conducting risk assessments and determining management strategies, food businesses must go beyond labeling controls as CCPs or PRPs. We need a broader, risk-based Food Safety Plan mindset. In cases like this, pre-cooked, hydrated components used in RTE foods inherently require elevated scrutiny not because something necessarily failed, but because the risk profile demands it. A broader system means: • Strong supply-chain preventive controls for high-risk ingredients.HACCP often treats supplier controls as PRPs. A Food Safety Plan treats high-risk incoming ingredients as Supply Chain Preventive Controls, applying CCP-equivalent rigor. • Sanitation Preventive Controls when the hazard is environmentalWhen environmental contamination is a reasonably foreseeable hazard, sanitation is no longer “just a PRP”, it becomes a managed preventive control. • Allergen Preventive Controls where cross-contact risk is significant.When severity is high, allergen management requires structured and robust preventive controls. • Verification beyond documentation.Does your supplier have validated preventive controls?Are you reviewing real performance data and environmental monitoring trends — not just Certificates of Analysis? • Environmental monitoring expectations at supplier level, where relevant. • Risk-based intensity of oversight proportional to severity. • and more…. Food Safety Plans change how we think. They shift us from diagram compliance to hazard-control strategy design. And that shift is not theoretical. It is necessary. If you manufacture or brand RTE foods, your strongest critical control may ALSO sit at your supplier’s site. I have prepared a very simplified illustration showing how HACCP and a Food Safety Plan approach may differ, both at the level of the pre-cooked pasta manufacturer and at the level of the brand owner using it in finished RTE products. Have a look at it here: If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, Food safety and trade, FSMA
Tagged under:
#foodsafetyplan, #fsp, #HACCP, #riskmanagement, Food Safety, Regulatory compliance, Risk assessment
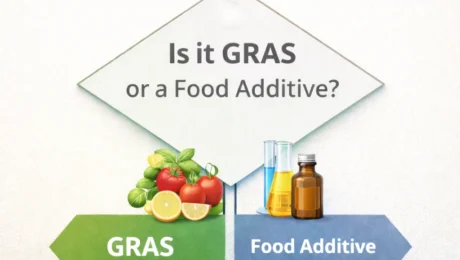
EU Compliance Is Not FDA Compliance: What Ingredient Status Means for US Market Access
Thursday, 29 January 2026
by Dr Faour Klingbeil
If raw materials aren’t fully FDA-compliant but EU compliant, can we still sell the product in the US ? This question comes up often with complex formulations. From an FDA perspective, the logic is simple. Pointing to approval in other jurisdictions, relying on a long history of use elsewhere, or assuming that a low use level or a so-called “minor” ingredient will be overlooked is not a good argument or approach to access the US market. These factors may feel reassuring, but they do not establish legal status under FDA rules. What does work is a disciplined regulatory approach by mapping each ingredient and sub-ingredient to its US regulatory status, verifying that the intended use levels and conditions of use are covered, and reformulating where no valid approval or GRAS pathway exists. I have prepared this illustration that summarizes the decision logic used to determine a substance’s regulatory and legal status, and shows where compliance ultimately leads to the need for food additive authorization. If you find it useful and choose to share it, attribution would be appreciated. If you have questions, feel free to reach out anytime. We support food businesses with food labeling compliance, risk assessment and management, and food system compliance with the FDA. If you have questions about what FDA registration means for your facility, which regulations apply to your operation, or how to design compliant food labels and build a compliant HACCP and Food Safety Plan for the US and EU market, DFK Safe Food Environment provides regulatory consulting, FSPCA training, and FSMA compliance support for domestic and international food businesses.
- Published in FDA Compliance, FSMA